|
Hydrogen attached to oxygen makes it an obvious candidate for hydrogen bonding. When Human safety with these acids arises, it does not matter which has lower or higher bond strength.Ĭommercially H2SO4 is 98% acid and HCl is 35% acid. That makes HCl stronger than H2SO4 as a molecule.ģ5% HCl vs 98% H2SO4: Which is more harmful to human safety Lower bond dissociation enthalpy of the H - Cl bond makes it lose proton more easily than H2SO4. In HCl, the bond dissociation enthalpy is 427 kJ/mol whereas in H2SO4, where the O - H bond breaks, it is 467 kJ/mol. The strength of an acid as a molecule is its ability to lose a proton, H+ ion in water. HCl vs H2SO4: Which is stronger? 35% HCl or 98% H2SO4? This makes KOH losing OH- ion more easily than Na and a stronger base. Therefore, K is more electro +ve /reactive than Na. 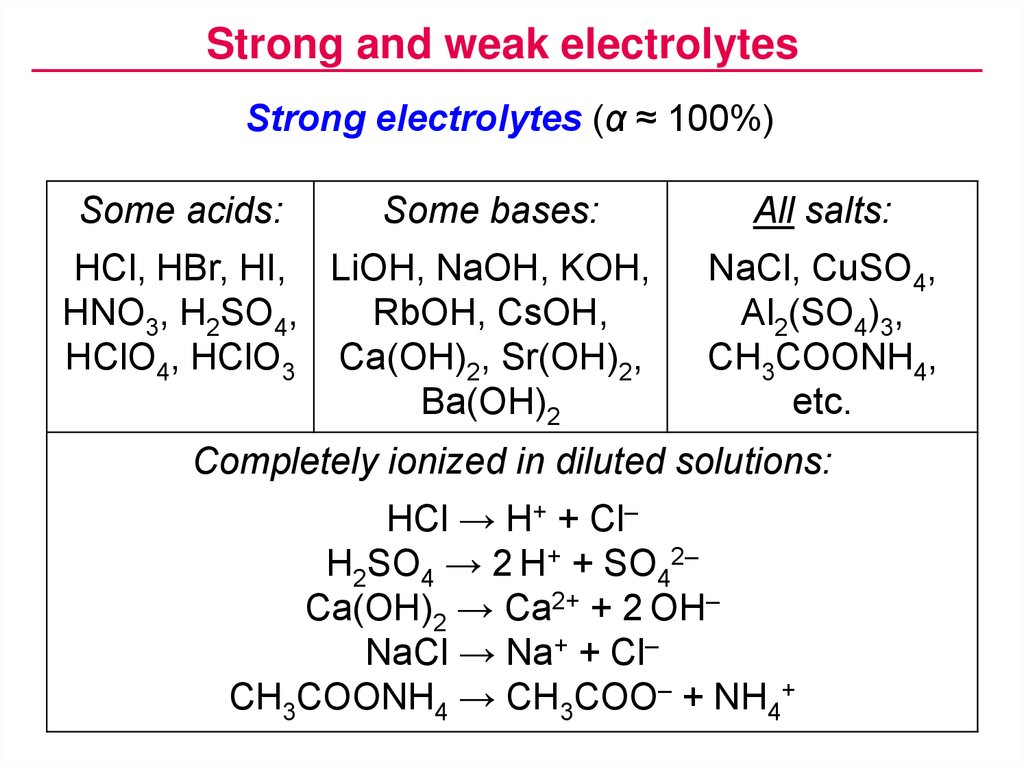
The ionization energy of Na is 495.8 kj/mol vs K has 418.8 kj/mol. This makes K ionizes more easily than Na. A larger number of inner electrons in K produces larger electron to electron repulsion to outer valency electron making it less attracted to protons in the nucleus and thus outer valency electron of K gets pushed out more easily and gets ejected out. K has one full extra energy subshell because of its higher atomic number 11 vs 19. While both have one electron in the outer valency shell. The electron configuration of K is 2,8,8,1, there is a total of 19 electrons spread over four energy subshells. The electron configuration of Na is 2,8,1, there is a total of 11 electrons spread over three energy subshells. In a group in the periodic table, an increase in the size of elements outweighs increasing nuclear charge. In the periodic table, Na and K both belong to group 1. Na atomic number is 11 while K's atomic number is 19. The question is which is stronger? The answer is one that loses –OH ion quicker in water. 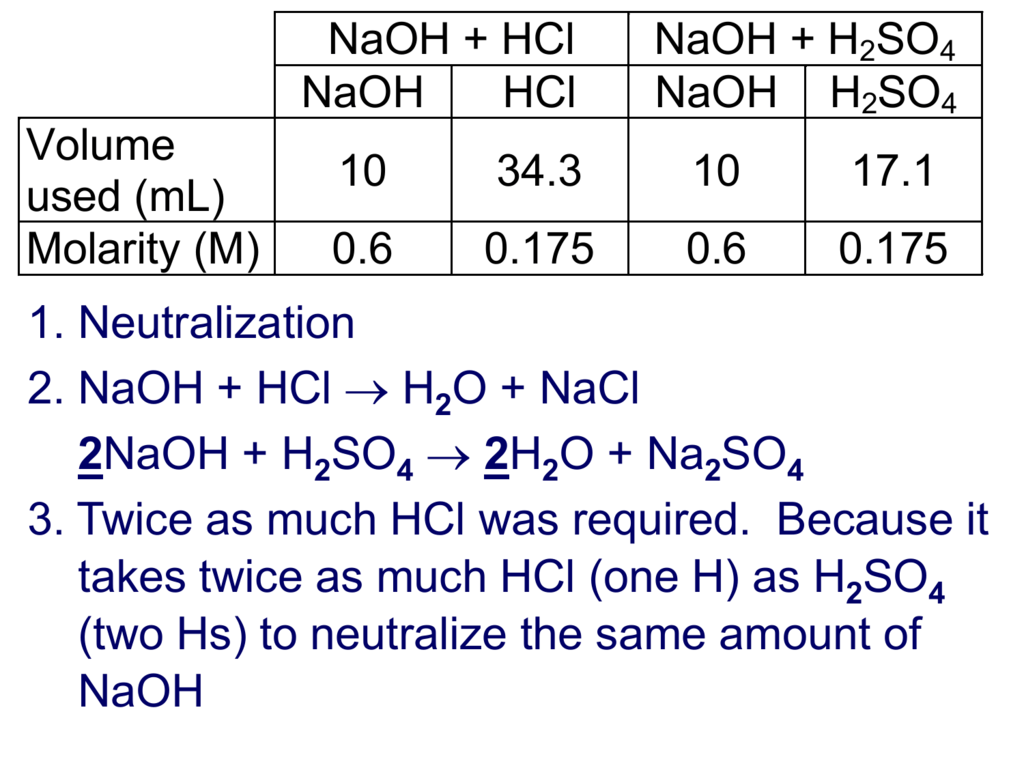
Ammonia is a good example of a weak base. In contrast, a weak base only partially dissociates into its ions in water. NaOH vs KOH: which is stronger as commercially available solid or 45-50% liquid?Ī strong base is a base that is completely dissociated in an aqueous solution like NaOH and KOH. Consequently, the pH of, for example, a 0.1 M solution of NaOH should be the same as a 0.1 M solution of KOH. In either case, they dissociate in water completely and they both produce the same base, OH. Sodium Hydroxide is more commonly known as lye or caustic soda where Potassium Hydroxide is known as potash.īoth KOH and NaOH are fully ionic strong bases.Īt equivalent concentrations, there is no difference in base strength when they exist as a dilute solution. They are both a white, strongly alkaline, corrosive solid or powder. 
They are the most chemically similar to hydroxides. Sodium Hydroxide (NaOH) and Potassium Hydroxide (KOH) are almost interchangeable.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
